December 19, 2019
by Andreea Sterea
Revolutionary CRISPR-based gene editing system disrupts the function of a bacterial gene that confers antibiotic resistance to humans.
What is the CRISPR-Based Pro-AG System?
A mixed research team from UC San Diego School of Medicine and the Division of Biological Sciences has been working with CRISPR technology to create a new gene-drive system. The Pro-AG system (“pro-active” genetic system) targets those genes that render bacteria antibiotic-resistant. As we already reported, antibiotic resistance is one of the biggest health threats we face today.
The Pro-AG system’s foundation is represented by a modification of the standard CRISPR-Cas9 technology of gene editing in DNA. The research team worked with E. coli. They managed to disrupt the way a bacterial gene works in conferring antibiotic resistance. One of the thorniest issues the scientists tackled is the ability of bacteria to transfer antibiotic resistance among themselves. As we know, the bacterial cells’ ability to pass antibiotic resistance to each other makes treatment one of the biggest challenges in medicine.
In essence, the Pro-AG system uses a cut-and-insert mechanism to alter the antibiotic resistant genes’ activity. It seems to have greater efficiency than current cut-and-destroy mechanisms.
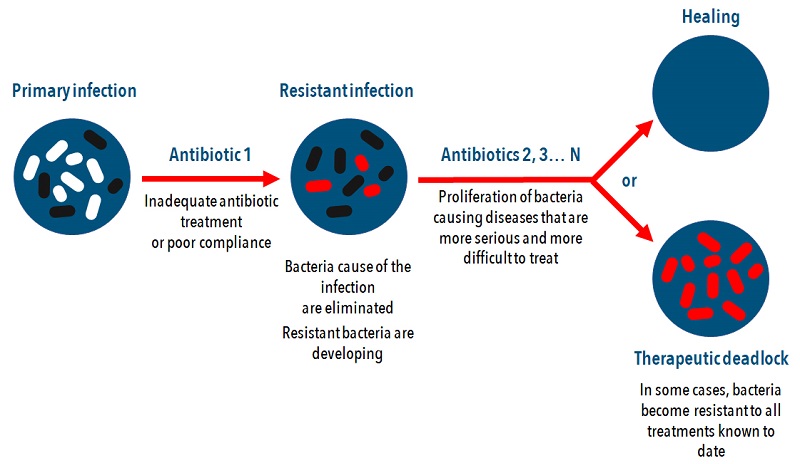
The Effectiveness of the Novel Pro-AG Technique
The findings, published in the journal Nature Communications, show enhanced effectiveness of the Pro-AG system. The scientists proved that this novel method worked extremely well in experimental cultures of bacterial cells containing genes known to offer antibiotic resistance to ampicillin. The Pro-AG method relies on the self-amplification of its editing features. It increaseas its own efficiency via a positive feedback loop mechanism. The result is that now scientists can insert customized genetic payloads into specific targets with high-magnitude precision.
The Pro-AG system is not ready yet to use on humans. However, it shows incredible potential in the treatment of patients with chronic bacterial infections. Moreover, the science team also emphasizes that the system is capable of editing its targets rather than destroying them.
It would mean that in the future, the system would enable the engineering or manipulation of bacteria for a wide area of functions. Science could render such bacteria harmless or even recruit them for beneficial purposes in biotech or biomedical applications.
According to Victor Nizet, distinguished professor of Pediatrics and Pharmacy and the faculty lead of the UC San Diego Collaborative to Halt Antibiotic-Resistant Microbes (CHARM),
“A human delivery system carrying Pro-AG could be deployed to address conditions such as cystic fibrosis, chronic urinary infections, tuberculosis and infections associated with resistant biofilms that pose difficult challenges in hospital settings.“
What Can We Hope from the Pro-AG System?
To paraphrase distinguished professor Ethan Bier from the Section of Cell and Developmental Biology, this method might lower the prevalence of antibiotic resistance in the environment as a whole. Given the accuracy, precision, and efficiency of the method, scientists could spread the system across populations of bacteria to render them harmless. We may actually witness in our lifetime the demise of antibiotic resistant bacteria.
Want more science now?
Check out our news page where we post interesting studies and discussions (sometimes mocking them mercilessly) for more.
